
Chemistry deals with the study of the structures, physical properties, and chemical properties of material substances. All gases, liquids, and solids are different in terms of their composition. So, the classification of the matter is very important.
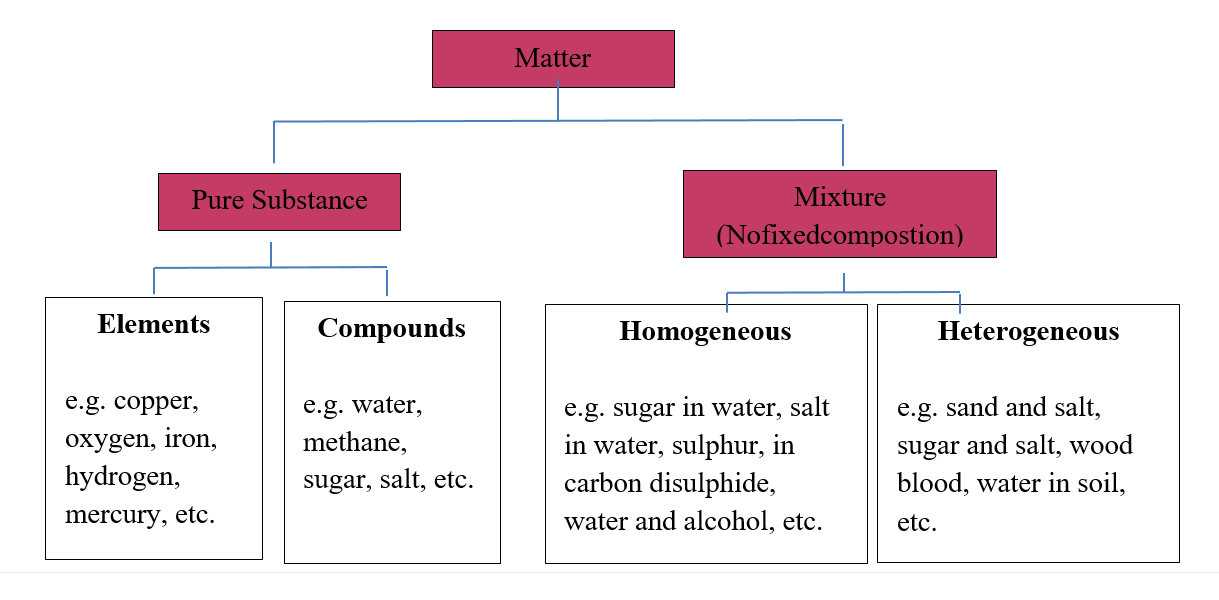
Matter is classified into three categories:
Classification of matter
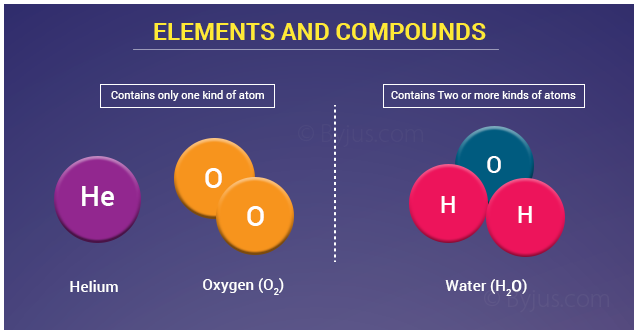
Elements:
Isotopes:
| Name of the element | Chemical symbol |
| Hydrogen |
|
| Boron | |
| Carbon | |
| Silicon | |
| Sodium | |
| Lead | |
| Platinum |