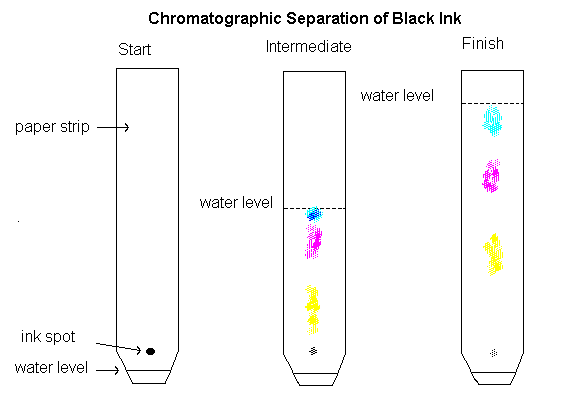- Chromatography is a technique of separating two or more dissolved solids which are present in a solution in very small quantities.
Principle:
- This separation is based on the fact that though two or more substances are soluble in the same solvent but their solubilities may be different.
- This method is based on the different properties of compounds in two phases: stationary and mobile phase.
- Based on this, chromatography can be classified into:
- Paper chromatography
- Column chromatography
- Thin layer chromatography
- Gas chromatography
Applications of Chromatography:
The technique of chromatography is used to separate.
- components of dyes
- pigments from natural colours
- amino acids
- sugars from urine samples
- drugs from blood
Purification by Crystallisation
- Crystallization is the process of cooling a hot, concentrated solution of a substance to obtain crystals.
- It is a better technique than ‘evaporation to dryness’ because of the following reasons:
- Some solids like sugar decompose or get charred on heating to dryness during evaporation. There is no such problem in crystallisation.
- The soluble impurities do not get removed in the process of evaporation. But such impurities get removed in crystallisation.
- Evaporation can be used with completely non-volatile solids.
Applications:
- Purification of salt that we get from sea water
- Separation of crystals of alum (phitkari) from impure samples
Separation by Distillation
- Distillation is the process of heating a liquid to form vapour, and then cooling the vapour to get back liquid.
- It is used for the separation of components of a mixture containing two miscible liquids that boil without decomposition and have sufficient difference in their boiling points.